+86 13438161196
+86 13438161196
Leave Your Message
-
 CONTACT NUMBER
CONTACT NUMBER -
 CONTACT NUMBER
CONTACT NUMBER -
 CONTACT NUMBER
CONTACT NUMBER



Nucleation is a fundamental process in various scientific fields, including materials science, physics, and chemistry. It refers to the initial stage of phase transition, where small clusters, or nuclei, form from a homogeneous phase. Understanding nucleation is critical for applications in industries such as pharmaceuticals, where it influences drug formulation, and in the production of advanced materials.
According to a report by the National Institute of Standards and Technology (NIST), nucleation plays a crucial role in determining the properties of materials, affecting everything from crystal size to the stability of emulsions. Research indicates that nearly 70% of new materials developed rely on controlled nucleation processes. However, the complexities involved are not widely understood, and mismanagement can lead to product failures or inconsistencies.
Many scientists struggle with the intricacies of nucleation. Entropy and energetics can be challenging concepts. Observations show that even experienced researchers face hurdles when predicting nucleation behaviors. This indicates a need for more accessible explanations and learning resources. This article aims to demystify nucleation, offering clear insights into its mechanisms and significance. By bridging the knowledge gap, we hope to empower both newcomers and experts in their understanding of this vital process.


Nucleation is a fundamental process in science. It serves as the initial step in the formation of new phases, such as crystals or bubbles. When a substance transitions, tiny clusters form. These clusters need to reach a critical size to grow further. This concept plays a crucial role in various fields, from meteorology to materials science.
Understanding nucleation requires recognizing its conditions. Temperature, pressure, and concentration often influence this process. For example, when water cools below freezing, ice crystals may form. However, this doesn't happen instantly. Sometimes, impurities in the water can help start the process more easily.
Yet, nucleation isn't always efficient. In some cases, the process can be inhibited. Factors like high viscosity or lack of surfaces for growth may prevent clustering. Reflecting on inconsistencies helps scientists improve methods to control nucleation. Fine-tuning these parameters leads to better outcomes in industries like pharmaceuticals and manufacturing.
Nucleation plays a crucial role in phase transitions and material science. This process marks the beginning of new phase formation, like ice crystals in water. Understanding nucleation is essential for developing materials with specific properties, such as strength, ductility, and thermal resistance.
In phase transitions, nucleation can be influenced by temperature and pressure. Research indicates that increased pressure can enhance nucleation rates. For instance, a study published in the Journal of Materials Science found that the nucleation rate in metals can double when pressure increases by 10%. This data highlights how small adjustments in conditions can lead to significant changes in material behavior.
Tips: When working with materials, consider environmental factors. They can dramatically impact outcomes. For instance, cooling rates during crystallization can alter microstructure, affecting the final material’s strength and ductility.
Nucleation is not perfect. It can lead to undesired qualities. In some cases, impurities act as nucleating agents, which may weaken the material. Monitoring these factors can help in achieving desired properties more reliably. Understanding these nuances allows scientists to create advanced materials for various applications, from aerospace engineering to biomedical devices.
This bar chart illustrates the average nucleation rates of different phase transitions in various materials. Nucleation plays a critical role in the formation of new phases, impacting material properties significantly.
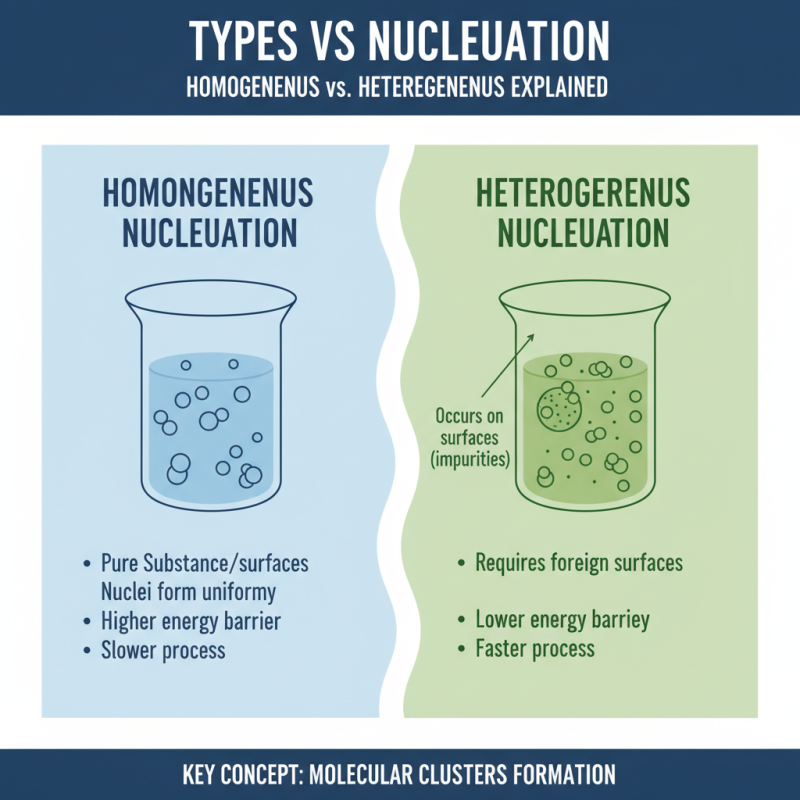
Nucleation plays a critical role in various scientific fields. It describes the initial process where clusters of molecules form. Understanding this concept is vital for fields like materials science and chemistry. Two primary types are homogeneous and heterogeneous nucleation.
Homogeneous nucleation occurs in a uniform environment. It involves droplets that form without any external influence. This process is rare and often requires specific conditions. Typically, it demands a significant degree of supersaturation. For example, when vapor cools rapidly, tiny clusters arise spontaneously. These clusters can grow into larger droplets or crystals.
Heterogeneous nucleation, on the other hand, happens on surfaces or interfaces. Impurities or container walls often facilitate this process. Here, the required energy barrier is lower. This distinction makes it more common in practical scenarios, such as cloud formation or ice crystallization. Heterogeneous nucleation is generally faster. The presence of nucleation sites accelerates formation, leading to quicker results. However, this raises questions about the impact of impurities. Are they always beneficial, or can they have adverse effects? Exploring these complexities is essential for advancing our understanding of nucleation.
Nucleation is a complex process influenced by various factors. Temperature plays a crucial role. Higher temperatures often increase molecular movement, which can either promote or hinder nucleation. Interestingly, cooling a liquid may lead to supercooling, where it remains in a liquid state below its freezing point. This state can create unstable conditions, significantly affecting nucleation rates.
Pressure is another vital factor in this equation. Increased pressure can compress molecules, speeding up the nucleation process. However, too much pressure can also disrupt the delicate balance needed for nucleation to occur. One might wonder how these factors interact. For instance, understanding the specific temperature and pressure conditions in different substances could lead to enhanced nucleation efficiency.
Supercooling adds another layer of complexity. When liquids are supercooled, they can resist crystallization for considerable time. Yet, this state is precarious. Under the right conditions, a single disturbance can trigger rapid nucleation. The challenge lies in predicting when that will happen. Therefore, exploring these factors provides valuable insights but also raises questions about their interplay and limits.
Nucleation is a fascinating phenomenon with wide-ranging applications across various industries. For instance, in crystallization, nucleation occurs when solute particles begin to cluster, forming a solid. This process is essential in pharmaceuticals, where controlled crystallization affects drug efficacy. When producing new medications, scientists carefully manipulate conditions for optimal nucleation. Small changes can dramatically impact the outcome, revealing the delicate balance in this scientific dance.
Cloud formation also illustrates nucleation's importance. Tiny water droplets form around dust particles in the atmosphere. This process is crucial for weather patterns. Understanding it can enhance weather forecasting and climate models. However, predicting cloud behavior remains a challenge. Factors like temperature and humidity introduce complexity, leading to unpredictable outcomes.
Despite these advancements, there is still much to contemplate about nucleation. Researchers often grapple with variables that influence nucleation rates. There’s a need for improved models to predict behaviors accurately. The quest to harness nucleation better continues, pushing the boundaries of what we know in both science and industry.
| Application Area | Description | Industry Impact | Nucleation Process Type |
|---|---|---|---|
| Crystallization | Formation of solid crystals from a homogeneous solution or melt. | Essential for pharmaceuticals, ensuring consistent drug quality. | Homogeneous and Heterogeneous Nucleation |
| Cloud Formation | Process where water vapor condenses into tiny droplets to form clouds. | Influences weather patterns and climate models. | Homogeneous Nucleation |
| Metallic Alloys | Nucleation of new phases that occurs upon cooling of metals. | Enhances strength and durability of metals used in construction. | Heterogeneous Nucleation |
| Ice Formation | Nucleation promotes the initial formation of ice crystals from water. | Affects ecosystems and temperature regulation in nature. | Heterogeneous Nucleation |
| Foam Production | Creation of stable bubbles through nucleation processes in liquids. | Used in packaging and insulation materials. | Homogeneous Nucleation |